Picture a future where obtaining a medication abortion is as simple as picking up a package from your local pharmacy. While this scenario remains hypothetical, it reflects a growing conversation among researchers and health professionals about the accessibility of abortion medication.
Dr. Daniel Grossman of the University of California, San Francisco, believes that such a future is within reach. A recent study published in JAMA Internal Medicine explores the potential for over-the-counter access to medication abortion, although there are currently no applications pending with the FDA to make this a reality. Dr. Grossman emphasizes, “It’s time that the general public understands that this could be a reality,” despite the legal restrictions present in more than a dozen states.
The Research Process
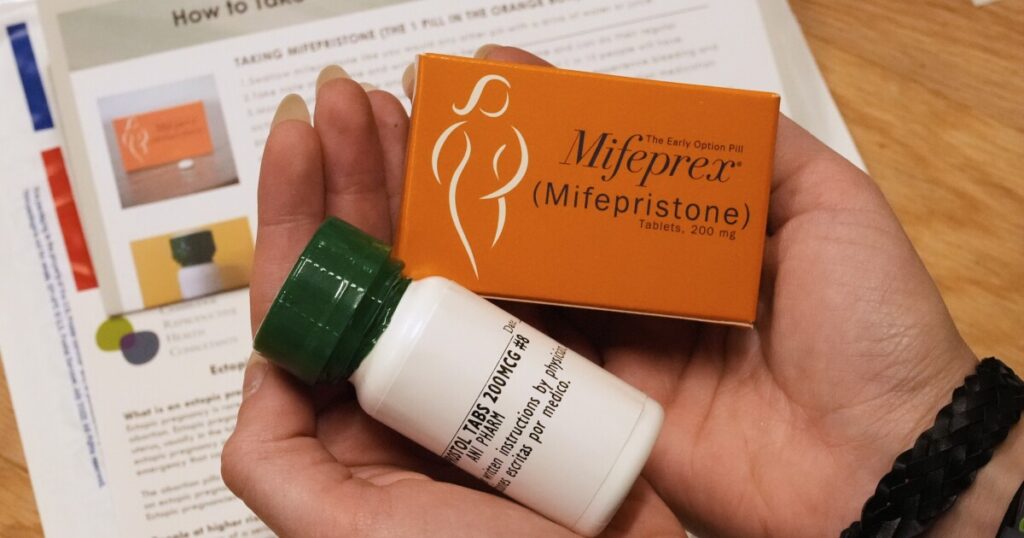
The study conducted by Dr. Grossman’s team involved 168 participants who were evaluated for their suitability for medication abortion. They were shown a prototype package named “MiMi,” which stands for mifepristone and misoprostol, the two drugs used in medication abortion. Participants self-assessed their eligibility based on the information provided, and their assessments were later compared to those of clinical professionals.
The findings revealed that 88% of participants correctly self-assessed their eligibility, either confirming or denying their suitability for the medications. Although Dr. Grossman acknowledges the small sample size and limited generalizability, he notes that the study is a step towards understanding the feasibility of over-the-counter abortion medication.
Broader Evidence and Commentary
In an accompanying commentary, Dr. Sonya Borrero from the University of Pittsburgh highlights the growing evidence supporting over-the-counter medication abortion. However, she cautions that the FDA’s decisions may be swayed by political pressures surrounding reproductive health.
Julie Maslowsky of the University of Michigan notes that the study’s scenario is not far from current practices, where medication abortion is often accessed through telemedicine. She argues that these drugs meet the FDA’s safety and efficacy criteria for over-the-counter availability, suggesting that such a shift would be an incremental change rather than a radical one.
Political Context and Challenges
The concept of an over-the-counter medication abortion kit faces significant political hurdles. Recent legislative actions, such as those by Senators Josh Hawley and Bill Cassidy, highlight ongoing opposition to mifepristone. The FDA’s impending safety review of the drug, spurred by anti-abortion groups, adds another layer of complexity.
Despite these challenges, Dr. Grossman remains optimistic about the potential for change. He draws parallels to the long journey of the over-the-counter birth control pill’s approval, emphasizing the importance of research in shifting public and regulatory perceptions. However, he concedes that the current political climate is not conducive to pursuing FDA approval for over-the-counter medication abortion at this time.
Future Directions
For over-the-counter medication abortion to become a reality, further studies are needed. These would include “actual use studies,” which involve tracking the real-world application of the drug, its appropriateness, and any outcomes. Julie Maslowsky notes that such studies are resource-intensive and complex but essential for regulatory progress.
—
Read More Michigan News